|
12/22/2023 0 Comments Water reactivity trend 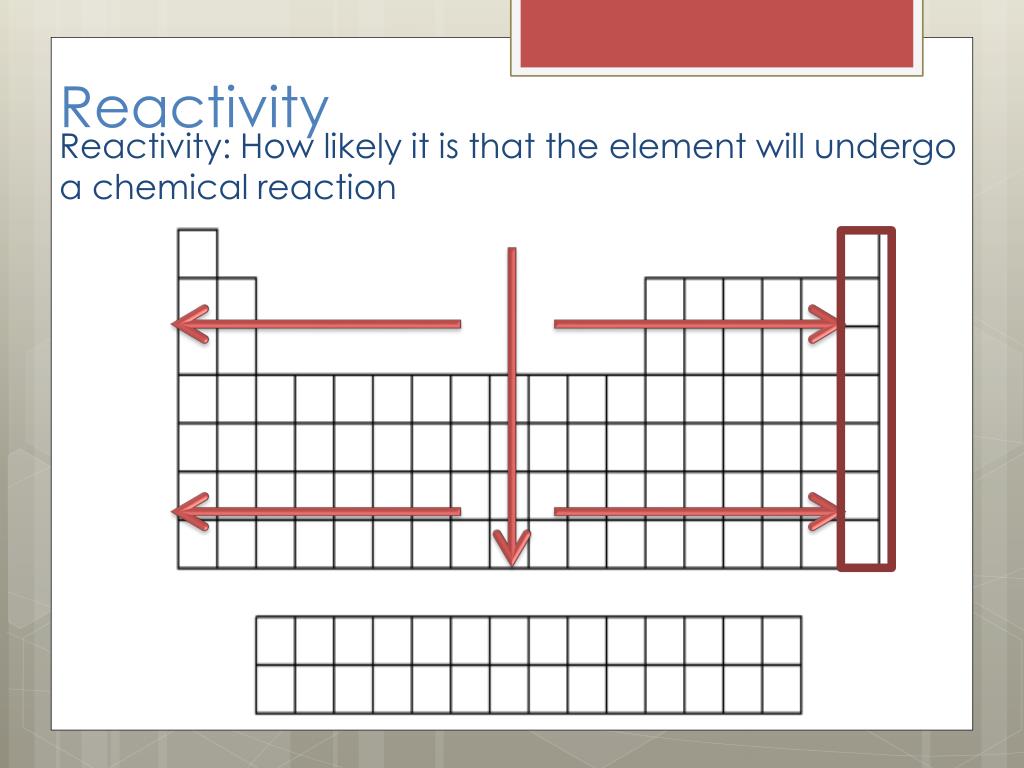
Knowing the atomization energy, the first ionization energy, and the hydration enthalpy, however, reveals useful patterns. The summation of these effects eliminates any overall pattern. The reaction becomes more vigorous as one moves from top to bottom in Group 1A: lithium sizzles fiercely in water, a small amount of sodium reacts even more. The extra protons in the nucleus are again screened by the extra layers of electrons. As the ions increase in size, the water molecules are farther from the attraction of the nucleus. These elements are abundant in the earth and thus acquire this name. The elements occurring in group 2 of the periodic table are alkaline earth metals. Group 1 and 2 metals are highly reactive and form hydrogen gas while the noble gases are so stable they do not react at all. Across the periodic table, different elements react differently to water. How Do Different Elements React with Water. However, the reactivity does increase down the group. This post will explore the reactivity of elements with water for Prelim Chemistry. On the other hand, reactivity in non-metals increase further right down a period, and further up a group. Reactivity of alkaline earth metals is such that they react with halogens to form halides, hydrogen to form hydrides, and acids to release dihydrogen. The trends in reactivity are not as clear in Group 2 as they are in Group 1. The reactivity of metals increases further left along a period, and further down a group. The hydration enthalpy is a measure of the attraction between the metal ions and lone pairs on water molecules. Reactivity is dependent on the classification of an element (metals and non-metals), as they both have differing periodic trends.Beryllium does not react at all, magnesium reacts very slowly whereas. The alkali metals, ranging from sodium to caesium, and the alkaline earth metals, ranging from magnesium to barium, are notable examples. The extra protons in the nucleus are screened by additional layers of electrons. The rate of reactivity with water of the elements increases as you move down the group. Water-reactive chemicals are those that undertake a chemical reaction with water on their own, due to the fact that they are naturally reduced in nature. 
The first ionization energy decreases because the electron being removed is more distant from the nucleus with each progressive atom. This post will explore the reactivity of elements with water for Prelim Chemistry.The delocalized electrons are further from the attraction of the nuclei in the larger atoms. This decreases as the size of the atoms and the length of the metallic bond increase. The atomization energy is a measure of the strength of the metallic bond in each element.There is no overall trend in the overall reaction enthalpy, but each of the component input enthalpies (in which energy must be supplied) decreases down the group, while the hydration enthalpies increase: These values are tabulated below (all energy values are given in kJ / mol): Element 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
